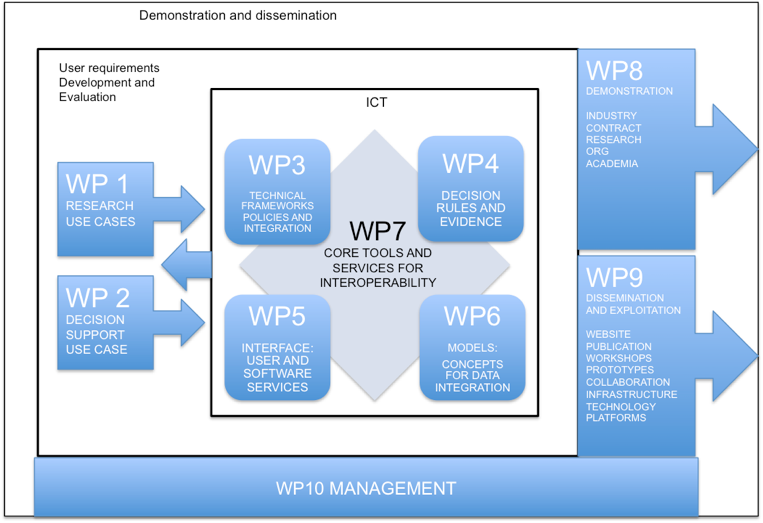
The basic concept of TRANSFoRm is that if data from research and clinical practice is to be integrated in support of patient safety, it is not sufficient to simply extract data from eHR systems, the data must be semantically enriched by the use of a live interface direct to the consultation. Similarly, automatic ‘translation’ of data is unreliable and a semantically aware workbench is needed to assist researchers in selecting appropriate codes. The core requirements for interoperability, semantically aware, dynamic interfaces and a rich ontology are common to all elements of research and knowledge translation, justifying their combination in this project and the ICT2009 work plan. TRANSFoRm will support clinical studies with potential patient safety value and directly support the use of evidence for diagnosis, reducing diagnostic error. Each WP will address one of the principal scientific objectives.
Clinical Use Cases: Three clinical use cases will run throughout the project, supporting the ICT work packages in three phases: use case development, testing and validation, and evaluation. The three clinical use cases are representative of the two phases of Translational Research [WP1] and Knowledge Translation [WP2].
User requirements Analysis: Two work packages develop the clinical use cases into highly detailed user requirements specifications in Legal, Privacy and Security [WP3], Decision Support rules [WP4].
Models and Standards: WP6 will take the detailed requirements and develop extensions to models and standards.
Development and evaluation: Two work packages will develop the software and services based on the requirements, models and standards. WP5 will develop dynamic, knowledge-driven interfaces for clinicians and researchers. WP7 will develop the integrated software and services for TRANSFoRm.
Validation, Demonstration and Dissemination: The software and services will be validated in the well-defined use cases [WP1 and WP2], with quality control and assurance monitored via WP3. WP8 will demonstrate the project results outside the existing use cases and WP9 will promote dissemination at all stages of the project. WP10 will hold the project management infrastructure and activities.